Many people like to think of periodontitis as a tooth- and gum-specific problem. If only it were that simple. Because once the immune system begins cannabilising the bone and tissue in the mouth, it struggles to stop there. That’s why patients with periodontitis are more likely to suffer a cardiovascular incident, why it can complicate diabetes management, lead to rheumatoid arthritis, and a host of other inflammatory diseases.
The bacterial trigger and the inflammatory cascade
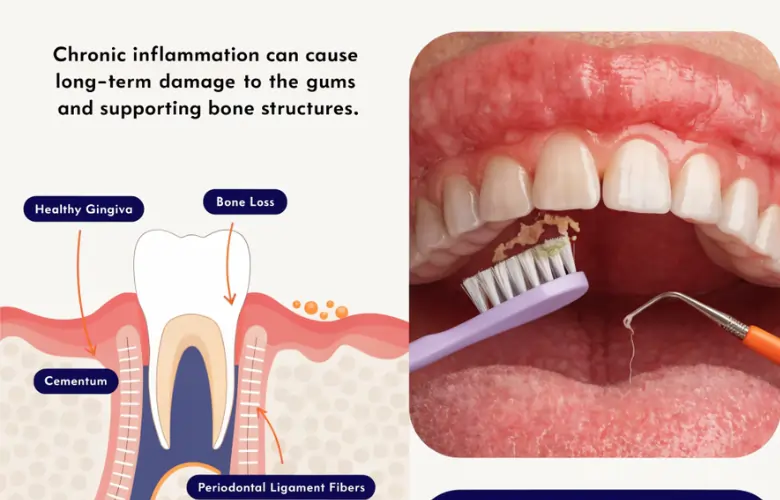
The disease process begins as the bacterial community in the mouth shifts from a healthy microbial population to one that is pathogenic. Mechanically the bacterium _Porphyromonas gingivalis_ localizes in the space between the tooth and the gum. This bacterium produces a number of toxins that stimulate the host’s immune response including the release of a group of signaling molecules by the body known as cytokines. These signaling molecules are pro-inflammatory, which in the short term is meant to keep the load of “infectious” bacteria in check. The long-term activation of the immune response is not turned off in the absence of the elimination of the bacterial “foe.”
The first to suffer from this attack are the tissues surrounding the tooth. The body’s immune attack on the bacteria starts to break down the alveolar bone, showing the body’s own friendly-fire casualties. Simultaneously, the ulceration-induced recession of the gingival tissue creates the pocket for the bacterium to live in, hidden from the thoroughfare of mouthwash, floss, and toothbrush. Left unmanaged, the bacterium will continue to quietly proliferate in its niche, nourished by the host and the waste byproducts of the body’s own attempts to eliminate and contain the invading cells. The more this “foe” and its byproducts multiply, the more the immune system is activated.
When home care isn’t enough
Brushing and flossing are pretty good for controlling biofilm at and above the gumline. They’re not good at controlling subgingival plaque once pockets get deeper than 4mm. At that point, the colonies of bacteria that have set up camp on a root surface are pretty well isolated from anything a toothbrush might achieve – and those colonies are what’s stimulating the inflammatory response.
That’s what scaling and root planing is for. The process removes biofilm and calculus from root surfaces below the gumline in order to reduce the bacterial load and give the immune response a chance to die down. It’s not a cure, but it is the critical first step in managing a state of chronic inflammation.
This is where general dental care tends to reach the end of its capacities. Patients with pockets deeper than 4-5mm, patients with systemic conditions that are associated with inflammation, or patients with radiographic evidence of bone loss should really be under the care of a dentist who specializes in inflammation, not just their local general dentist. A Melbourne periodontist, for instance, will be trained to conduct an overarching inflammatory assessment which takes both the oral and systemic condition into account – this is not something your day-to-day cleaner is really set up to do.
The bidirectional link with systemic disease
The relationship between periodontitis and diseases like Type 2 diabetes isn’t a one-way street. Elevated blood glucose hinders the repair of damaged gum tissue, providing a refuge for infection. Meanwhile, the inflammatory state caused by active gum disease represses the immune system’s ability to control blood sugar because systemic inflammation can prevent proper insulin signaling.
Comorbidities like obesity, diabetes or hypertension are all mainstay medical issues in Western society, and all three relate to periodontitis via common inflammatory pathways. Patients with multiple chronic diseases often exhibit more severe periodontitis and a suboptimal response to standard therapy. Oxidative stress further enhances this vicious cycle. The imbalance between free radicals and antioxidants in inflamed tissue can amplify the breakdown of the gums and interfere with the repair process on a cellular level.
Almost 47.2% of adults aged 30 and older have some form of periodontal disease. The percentage increases to 70.1% among adults 65 and older. Given the prevalence of periodontitis, the overall disease burden related to the systemic consequences remains underestimated outside the specialist field.
Lifestyle factors that accelerate the damage
Cigarette smoking comes up in every conversation about lifestyle modifications and periodontal disease, because it is a primary modifiable risk factor, if not the primary one. It promotes dysbiosis of the gingival oral microbiome and suppresses the immune system’s response to the inciting pathogens. In other words, it makes the wrong bacteria grow in your mouth and then sabotages your immune system’s efforts to fight them.
Smoking not only makes you more susceptible to periodontal disease; it also masks the clinical signs and symptoms. Most notably, nicotine constricts blood vessels, which supplies the gingiva with the oxygen and nutrients needed to form a functional immune response to the bacteria that cause the disease. Constricted blood flow means your body can’t fight the infection as well. It also can’t move immune cells to the site of infection as effectively, nor can it clear away the waste products from their battle as efficiently. Additionally, nicotine decreases the underlying recellularization process of the body. This repair and regeneration activity cannot function optimally due to a lack of blood flow.
Treating gum disease as what it is
How the message is received is crucial. For example, a patient will be more inclined to perceive treatment for his bleeding gums as important if he understands that chronic inflammation is making it difficult for him to manage his blood sugar or that his inflamed gum tissue is undermining his efforts to count calories and lose weight than if he has only been told that he needs to floss or get his teeth cleaned more often.